 Even if not flammable, it could collect in the lower floor or level of a confined space and displace air, possibly presenting an asphyxiation hazard to individuals entering the lower part of that space. Density is almost identical to specific gravity, but not quite. Density is very slightly less than specific gravity. The density has implications for container storage and personnel safety-if a container can release a dense gas, its vapour could sink and, if flammable, collect until it is at a concentration sufficient for ignition. However, because the density of pure water is so close to 1 (0.9976 grams per cubic centimeter), specific gravity and density are nearly the same value so long as the density is given in g/cc. With this definition, the vapour density would indicate whether a gas is denser (greater than one) or less dense (less than one) than air. 
Below is a table of units in which density is commonly expressed, as well as the densities of some common materials. There are exceptions however, such as water's density increasing between 0C and 4C. The output density is given as kg/m, lb/ft, lb/gal (US liq. The calculator below can be used to estimate the density and specific weight of liquid ethanol at given temperature. This can be seen by dividing the molecular weight of Acetone, 58.1 by that of air, 28.97, which equals 2. Increases in temperature tend to decrease density since the volume will generally increase. Tabulated ethanol density specific weight at given temperature and pressure (SI and Imperial units) as well as density units conversion are given below the figures. That means acetone vapour is twice as heavy as air. Ice, water’s solid phase, is more buoyant, so it forms at the surface of water bodies and freezes downward. For example, acetone has a vapour density of 2 in relation to air. Water’s density is greatest at about 4 ☌ (39.2 ☏), in the liquid phase. For this use, air has a molecular weight of 28.97 atomic mass units, and all other gas and vapour molecular weights are divided by this number to derive their vapour density. In many web sources, particularly in relation to safety considerations at commercial and industrial facilities in the U.S., vapour density is defined with respect to air, not hydrogen. Vapour density is a dimensionless quantity. 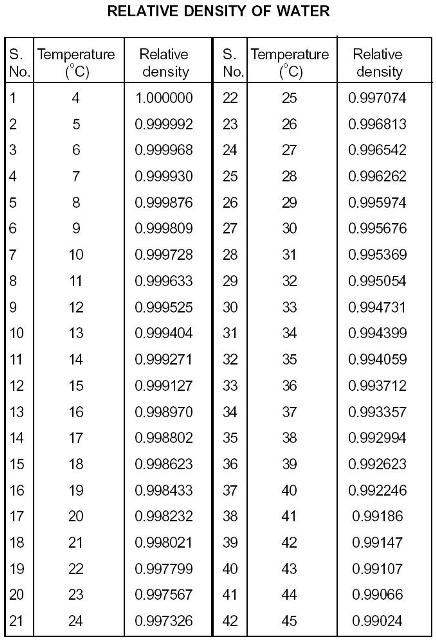
(and thus: molar mass = ~2 × vapour density)įor example, vapour density of mixture of NO 2 and N 2O 4 is 38.3. vapour density = molar mass of gas / molar mass of H 2 vapour density = molar mass of gas / 2.016 vapour density = 1⁄ 2 × molar mass This simplest hydrogen chalcogenide is by far the most studied chemical compound and is described as the 'universal solvent' for its ability to dissolve many substances. At room temperature it is a tasteless and odorless liquid, nearly colorless with a hint of blue. Vapour density = mass of n molecules of gas / mass of n molecules of hydrogen gas. Water ( H 2 O) is a polar inorganic compound. It may be defined as mass of a certain volume of a substance divided by mass of same volume of hydrogen. Vapour density is the density of a vapour in relation to that of hydrogen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
